PlantStrength – Background
The plant pathogen Fusarium langsethiae produces highly potent mycotoxins (HT-2 and T-2). These are at high levels in some Norwegian oat lots, and may constitute a considerable risk for food and feed safety. Research of this host pathogen system has come into focus only recently and many questions regarding the biology and epidemiology of F. langsethiae and the interaction with the host phenology remain open. Understanding these interactions is instrumental in order to develop measures directed towards minimising infection and contamination of cereals (Imathiu et al. 2012/Journal of Phytopathology).
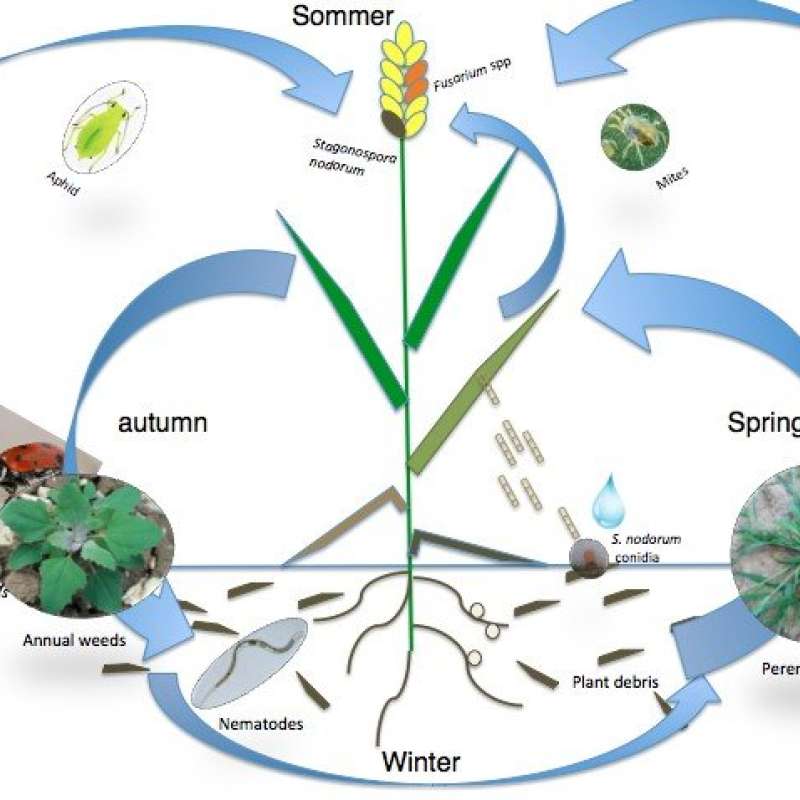
The plant pathogen Stagonospora nodorum can cause 20-30% yield loss and significantly reduces yield quality in Norwegian spring and winter wheat (Brodal et al. 2009/Bioforsk FOKUS). Cultural management, such as crop rotations and ploughing can reduce the local inoculum source. However, there is evidence that sexually produced ascospores can travel long distances and invade a field with no local infection source present (Sommerhalder et al. 2010/Phytopathology). Understanding the abiotic factors influencing ascocarp development and ascospore production, release, transport and survival, will help us to better predict S. nodorum epidemics and target management practices to the periods of highest infection risks.
The two most important aphid species in cereals in Norway and elsewhere are the Bird cherry-oat aphid (Rhopalosiphum padi) and the English grain aphid (Sitobium avenae). These may cause significant economic damage and treatment threshold levels of these aphids have been established (Klingen et al. 2009/Bioforsk FOKUS). However, these threshold recommendations are not always followed, leading to redundant pesticide use. The entomphthoralean fungi Pandora neoaphidis and Entomopthora planchoniana have been found to cause epizooticks in R. padi and S. avenae (Nilsen 2002/The Royal Veterinary and Agricultural University, Copenhagen, Denmark. PhD Thesis). Utilizing these fungi in biological control might constitute a valuable opportunity to reduce insecticide use in cereal production. However, predictions of potential epizooticks by entomopathogenic fungi should only be based on solid knowledge about the complexity of factors affecting pathogen activity and pest population growth (Klubertanz et al. 1991/Environmental Entomology). These factors and their interactions are not well known (Klingen & Westrum 2007/Biological Control).
The yield loss in grains caused by the perennial grass weed Elymus repens has been estimated to be 10-40% (Melander 1994/Weed Research; Berge, unpublished data). E. repens is controlled by the herbicide glyphosate and/or mechanically. Optimal effect of these direct control measures depends on a close alignment between timing and the period of highest plant susceptibility. The period of highest susceptibility is a function of the phenological stage and abiotic factors. Because E. repens grows in patches within fields, a sustainable approach is to apply the control measures site-specifically. This requires efficient detection or mapping methods, and sensor-based methods are underway. Therefore, both the traditional measure on thermal time for each new leaf to appear (‘phyllochron’) and a novel measure, based on leaf ground coverage estimated with automatic image analysis, will be explored.
The alien, invasive annual plant Impatiens glandulifera produces a large amount of seeds and threatens the native species composition in Norway and Europe (Gederaas et al. 2012/Fremmede arter i Norge- med norsk svarteliste). It has been observed in spring cereal fields in SE Norway. A cost-effective management strategy to eradicate the species is required. As for E. repens, the basis is a valid phenological model for the species. Since it reproduces by seeds, the time required to flower is of particular interest (the ‘phenjuv’ parameter, cf. WTDB/Endure database), as well as whether different ecotypes, or the recommended cutting for populations near waterways, affect the flowering phenology.
Pest arthropods and their natural enemies (beneficial fungi, predators and parasitoids) are known to interact with weeds, plant pathogenic fungi and fungicides in ways that may enhance or decrease their efficacy as natural enemies or pests (e.g. Klingen & Westrum 2007/Biological Control; Asalf et al. 2012/Biological Control). Powell et al. (1986/Crop Protection) showed that herbicide applications reduced natural enemies of aphids, particularly polyphagous predators and entomopathogenic fungi. They suggested that the higher prevalence of entomopathogenic fungi on the pest aphid S. avenae in the weedy plots was caused by a higher relative humidity (RH) or that the weeds were associated with other aphid species infected by entomopathogenic fungi, which spread to S. avenae. RH is also important for the development of plant pathogenic fungi, such as S. nodorum and F. langsethiae. Density of different weed species could influence the epidemic development of aphids, entomopathogenic and plant pathogenic fungi by their effect of RH and light perception (e.g. de Castro et al. 2013/ Journal of Invertebrate Pathology). The weed flora may be manipulated both qualitatively and quantitatively to manage some parts of the cereal pest complex without affecting crop yield. Both fungicides and herbicides do not only affect their target organism, but might also influence beneficial entomopathogenic fungi (Klingen & Haukeland 2006/Progress in biological control, Springer). Interactions between weeds, aphids, their natural enemies, wheat pathogenic fungi, and their hosts are complex, and therefore mostly studied separately. However, managing plant pest complexes more effectively requires a system approach, where the factors influencing each pest, pathogen or natural enemy will be studied in combination.
In comparison to pure statistical models, computer simulation models can capture such complex interactions. Computer simulation models constitute a key component of modern forecasting systems and decision support systems for pest control. A computer pest model is an instrumental summary of a pest in its environment, written in the language of mathematics and logic. Generally, such models are systemized biological knowledge, which usually takes biological observations, climate data and weather prognoses as inputs. The purpose of such models in our project is twofold: i) to optimize the timing and dosage of pesticides for sustainable and profitable crop protection and ii) to serve as a research tool for hypothesis testing in complex systems. In our project, we will utilize the power of simulation models to understand the complex nature of the interacting biotic and abiotic factors in the oat-Fusarium system and the cereal pest syndrome (see below).
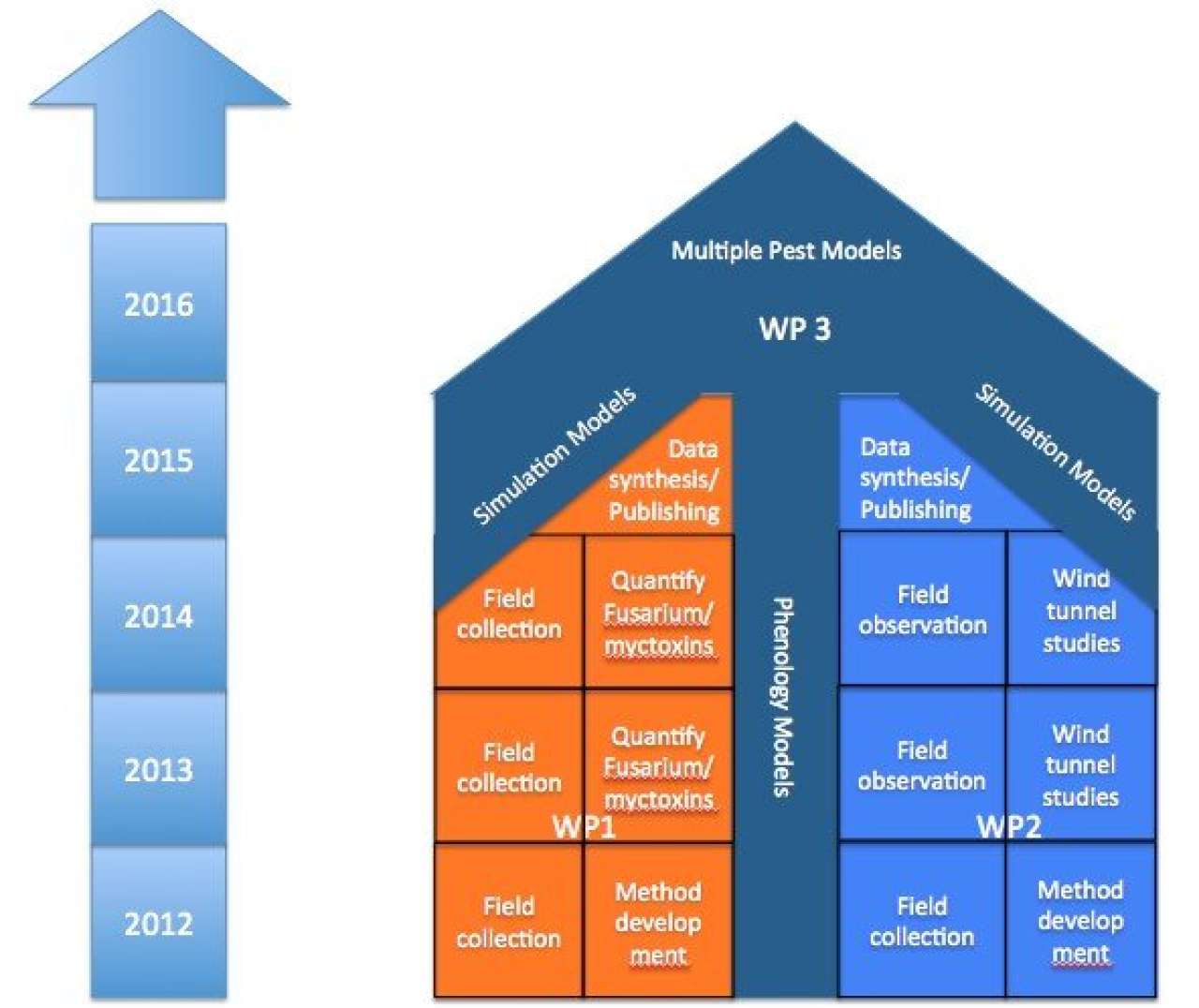
Workpackages
1) Fusarium-oat system
2) Cereal pest syndrome
3) Computer simulation models
Scientific methods
Molecular (qPCR) and morphological methods for identification of Fusarium in grains, plant debris, insects and soil as well as ELISA methods for rapid mycotoxin analysis (DON and HT2+T2) in cereals, will be validated and implemented. Greenhouse and laboratory experiments will be conducted to study interactions between oats, F. langsethiae and potential vectors.
Methods to study perithecia development and ascospore release in S. nodorum using opposite mating types under controlled conditions will be developed to study the effect of abiotic factors, such as light quality, intensity and cycles, in small wind tunnels.
Controlled laboratory/microcosm experiments on abiotic factors (temperature, RH, light) important for the sporulation of aphid pathogenic fungi will be designed. Field collection and observation of aphids and their natural enemies from the cereal field and the border vegetation will be done to obtain information on the actual insect pest and natural enemy situation in Norwegian cereal fields.
Images of plots containing E. repens and phenological observations (extended BBCH scale, cf. Hess et al. 1997/Weed Research) of individual E. repens shoots will be taken in cereal stubble fields. Automatic image analysis software (e.g. Berge et al. 2012/Computers and Electronics in Agriculture) will estimate the ground coverage of E. repens in the images. To find the best ‘phyllochron’ parameter, temperature from soil, field layer and 2 m above ground and combinations with radiation will be tested. Phenological observations of I. glandulifera in pots and/or semi-field trials will be taken and weather data from the nearest weather station reported. To test the generality of the parameter ‘phenjuv’, 2-3 different ecotypes and one cutting (cut/not cut in mid-June) will be included.
Controlled laboratory experiments on the effect of selected abiotic factors on multi-trophic interactions between weeds, aphids, their natural enemies and plant pathogenic fungi in cereals will be conducted. The focus will be on microclimatic RH, pesticides and light.
Knowledge gaps on pest population dynamics and their interactions with other abiotic and biotic factors will be addressed by designing computer simulation models. These models will primarily be driven by daily meteorological and generated biological data. Designed computer models will be transparent and compatible with each other to allow merging them into complex crop/multi-pest model. The software Universal Simulator (Holst 2013/Ecological Informatics), STELLA and R will be explored for this purpose
Projects
Division of Biotechnology and Plant Health
PlantStrength – Strenghtening the basis of sound plant protection

Division of Biotechnology and Plant Health
SMARTCROP
Norway has implemented EU´s Directive on sustainable use of pesticides (2009/128/EC) which promotes the use of Integrated Pest Management (IPM) and alternative approaches or techniques (IPM tools) to minimize pesticide use. There is a lack, however, of proven and practical IPM tools for farmers to use, as well as a relevant policy for a successful implementation of IPM.
