Eva Farkas
Research Scientist
Abstract
presentation about circular economical projects in NIBIO
Abstract
Quantifying the impact of biochar on carbon persistence across soil textures is complex, owing to the variability in soil conditions. Using artificial soils with precise textural and mineral compositions, we can disentangle the effects of biochar from the effects of soil particle size. We can show that biochar application significantly reduces the early-stage carbon mineralization rates of plant residues in various soil textures (from 5 % to 41 % clay) but more significantly in sandy soils. Clay and silt particles alone also reduce C mineralization, but the magnitude of the changes is negligible compared to the impact of biochar. This finding suggests that biochar can compensate for the lack of clay in promoting C persistence in soil systems. This short report contributes substantially to understanding soil texture and biochar application interactions.
Abstract
No abstract has been registered

Division of Environment and Natural Resources
AgroComposit
AgroComposit: Biochar-compost composites for supporting site-specific soil agro-ecosystem functions and climate change mitigation

Division of Environment and Natural Resources
AgriCascade
Cascading recycling of organic N-sources with next-generation biochar fertilizer for Norwegian agriculture
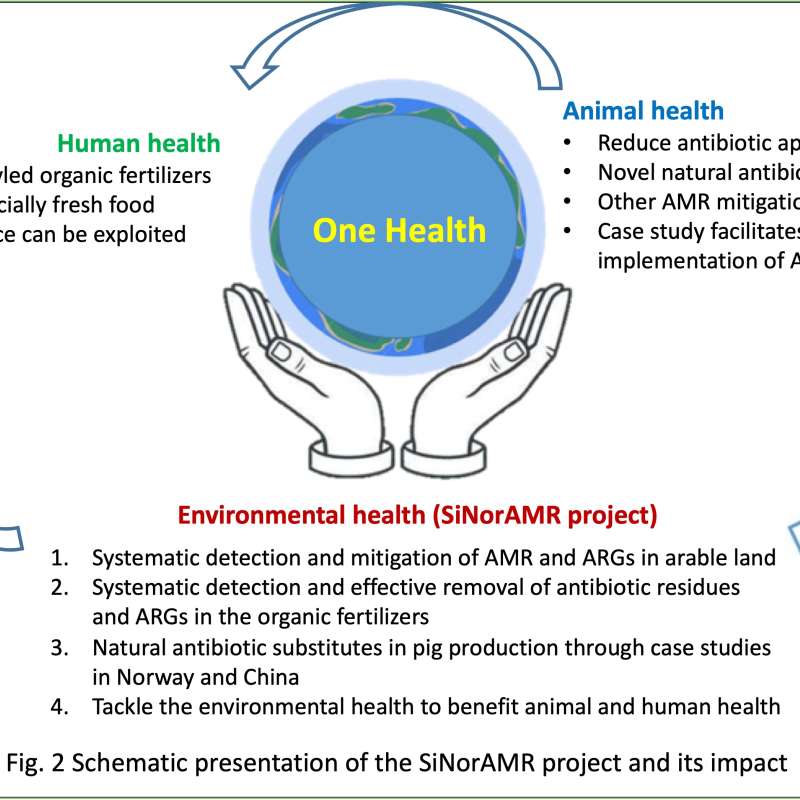
Division of Environment and Natural Resources
SiNorAMR
Full title: Collaborative and Knowledge-building Project Collaborative Project Systematic detection and mitigation of antimicrobial resistance in soil environment and animal health contributing to human health (SiNorAMR)
